ACCES 2024 Conference Cancellation
Dear Members,
We regret to inform you that due to some unforeseen economical issues with the location, we must announce the cancellation of the ACCES conference scheduled for this year in Halifax. However, we are excited to share
Winter 2024 ACCES Update
Our President, Gary Slaunwhite, has announced his resignation as President of ACCES. In response, Peter Butt has been appointed as the acting President by the Board of Directors to guide the organization through this transition.
Message from the President, October 2023
October 2023
Well, here we are in the midst of our Fall season - my how the Summer flew by! I have a few exciting things to mention since our last newsletter.
We are currently gearing up for our ACCES26 Annual Atlantic Canada Clinical Engineering Society’s Educational Seminar and Trade Show taking place in 2024. Our ACCES26 chairman, John Inch ...
SNB CE Week Celebrations
CE WEEK CELEBRATION REPORT:
FOSTERING UNITY AND CREATIVITY THROUGH ENGAGING ACTIVITIES
By Sreekala Kuzhikkattipurath
During CE Week, our department organized a week-long celebration dedicated to Clinical Engineering (CE). From May 14th to May 20th, we came together to foster teamwork, ignite creativity, and honor our collective passion for the field. Let’s explore the highlights of this unforgettable event!
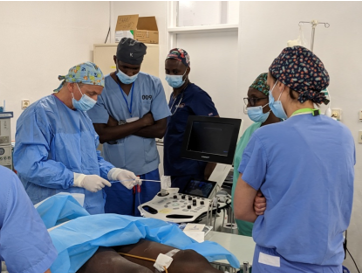
Spotlight Article – CR-OP’s Rwanda Mission – Part 1
Introducing CR-OP's Transformative Mission: A Beacon of Hope in Cardiac Surgery
In the heart of Halifax, Nova Scotia, a remarkable initiative known as CR-OP is quietly unfolding. This independent group of dedicated medical professionals hails from diverse corners of Canada and as far away as Poland...
CMBES Call for papers!
Dear Colleagues,
Welcome to this year's Canadian Medical and Biological Engineering Conference (CMBEC) and the Clinical Engineering Society of Ontario (CESO) 2024 event. This conference is a significant gathering for professionals in the field of biomedical engineering, and CMBEC46/CESO 2024 will take place in Toronto, Ontario from May 28 to May 30, 2024.
Nominate CE of the Year
Do You Know an Outstanding Clinical Engineering Professional?
We invite members of the ACCES community to submit nominations for the prestigious 2023 "ACCES Clinical Engineering Professional of the Year Award".
2023 Clinical Engineering Week Photo Contest Winner
In what has become a popular Clinical Engineering week tradition, departments from across Atlantic Canada came together to celebrate the National Biomedical and Clinical Engineering week held this past May. In celebration, ACCES once again held its annual photo contest ...
2023 Award Recipient
Congratulations to Phil Bradfield, esteemed member of our biomedical engineering community, on receiving the highly prestigious Lifetime Achievement Award!
Digital Marketing Sub Committee Call for Interest
Attention ACCES members! We're excited to announce the launch of our brand new Digital Marketing Subcommittee. Are you passionate about leveraging the power of digital platforms to elevate our organization's reach and impact? Join our committee and be at the forefront of driving our digital marketing initiatives.